Hello, Dear Readers!
Here is another post going into the highlights of one of our more recent papers dealing with dinosaur ichnology: the study of foot injuries in the fossil record!
Have you ever stubbed your toe? Pulled a muscle in your leg? Walked anywhere with a rock in your shoe? It doesn’t take major discomfort to figure out that foot and leg injuries can result in you walking “funny.” Perhaps you had to hop around on one foot for a bit. Maybe you could only take a certain length of step using your injured leg. All of the compensations that you make to avoid further or greater discomfort or pain have a good chance of being seen in your trackway. These modifications due to pelvic limb injuries also have a good chance of being preserved in the fossil record.
Tread Carefully
Of course, we have to be careful when looking at a fossilized trackway and seeing something “odd” about, for example, the gait of an animal. One aspect that can confuse people about vertebrate ichnology is that there is a HUGE amount of variation in how footprints are preserved and in how the animals themselves moved. All of that variation is perfectly normal. Dinosaurs (and any other vertebrate ambling over the landscape) are not metronomes. They are not robots. They will absolutely not take a step that is EXACTLY 345 cm long each and every time they step, or place their feet EXACTLY the same way every time. Some oddities in trackways are just that: oddities that are due to the natural variation in how a living, breathing, complex animal interacts with its environment as it moves from Point A to Point B.
In other words, when we look for phenomena that we can call pathologies, we are looking for repeated abnormalities in footprint shape and movement. This is the framework we used to review reports of fossilized footprints that preserve oddities that cannot be explained by poor preservation or an animal being an animal.
What Is An Ichnopathology?
When we discuss pathologies, or in this case ichnopathologies, we’re talking about trauma (bone or soft tissue) that would result in an animal walking differently than it would be expected to walk.
A pathology of the foot would result in direct preservation of the soft tissue and/or skeletal trauma that the foot experienced, such as a dislocated, broken, or amputated toe. A pathology of the lower (tibia and fibula, and muscles) and/or upper leg (femur and muscles) would result in a pace (a footstep) and stride (how the animal moves from right footprint to right footprint, or the “right-left-right” sequence of the trackway) that is different, such as a limp, shuffle, or foot drag, while the footprint itself may (but not always) look completely normal.
What Is Not An Ichnopathology?
There are footprint phenomena that are not ichnopathologies, no matter how strange they may look. Here are a couple of examples.
A. Missing Toes versus Natural Morphology
Here’s the danger of looking at isolated, singleton footprints. Let’s say you see a single footprint with a missing digit II (inner toe). Is this footprint the result of an injury, or is it the footprint of a dromaeosaur? It may be easy to see that the toe is missing, but looking at the trackway is essential in making an accurate interpretation of why that inner toe is “missing”.
Compare these two images:

The top image from Abel (1935) shows a large theropod trackway with a toe missing on only the right footprint. The bottom image from Xing et al. (2012) shows that there is a toe “missing” on both left and right footprints. Both trackways show this as a repeated occurrence. When we see a toe repeatedly missing from one foot only, that is a likely candidate for a pathology. When we see a toe missing repeatedly on both feet, it was likely never there to begin with, as is the case with dromaeosaurs. In fact, a consistently missing digit II is one of the synapomorphy-based characters we can use to confidently identify a trackway as belonging to a member of Paraves.
We have also observed dinosaur trackways where only one footprint shows a missing toe, while all of the other toes are more or less impressed. Those occurrences are most easily explained by preservation, rather than injury or anatomy: not every footprint within a trackway is going to be beautifully preserved.
B. Limping versus Laterality
Remember before when I said that animals aren’t metronomes? It is not uncommon for perfectly healthy animals to favor one limb over the other. This might result in a trackway that looks like it preserves a limp. However, “limping” is a loaded term: it implies that there was an injury or defect that caused the animal to walk the way it does. Data collected from emus (Dromaius novaehollandiae) shows that emus may take longer paces when stepping off with their right foot than if they step off with their left foot (McCrea et al., 2015). In other words, emus are right-handed, or right-footed. Ostriches have also been observed to be a bit right-footed (Bachiodonna et al., 2010). These irregular walking patterns aren’t the cause of injury, but rather because of laterality.
C. Anatomical Anomalies
I have a few bizarre requests for the Universe in terms of cool fossils to be found. One is polydactyly in dinosaurs, or even in a fossil felid trackway. When I was a child my family adopted a polydactyl cat (also known as a Hemmingway cat). Charlie’s hands sported two extra digits each, while his feet each had one extra digit. If Past Me had known Present Me was going to be this much of an ichnology geek, Past Me would have taken pictures of his footprints. No matter how many toes a cat has, there are still features on the foot (and the footprint) that would make it easy to identify it as a cat footprint, like the tri-lobed metatarsophalangeal pad.

Polydactyly is not as uncommon as one might think in the fossil record. Early tetrapods, specifically the early amphibians, that first started making their appearance on land in the Carboniferous had more than five fingers and toes on their hands and feet.
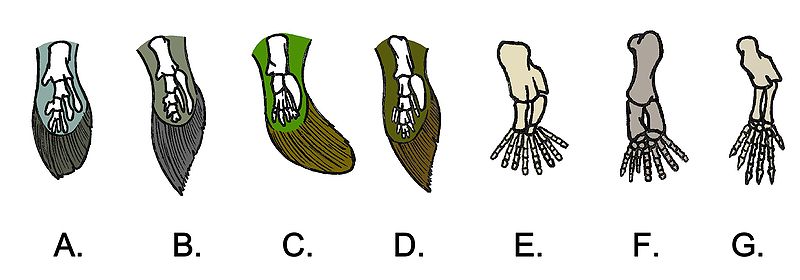
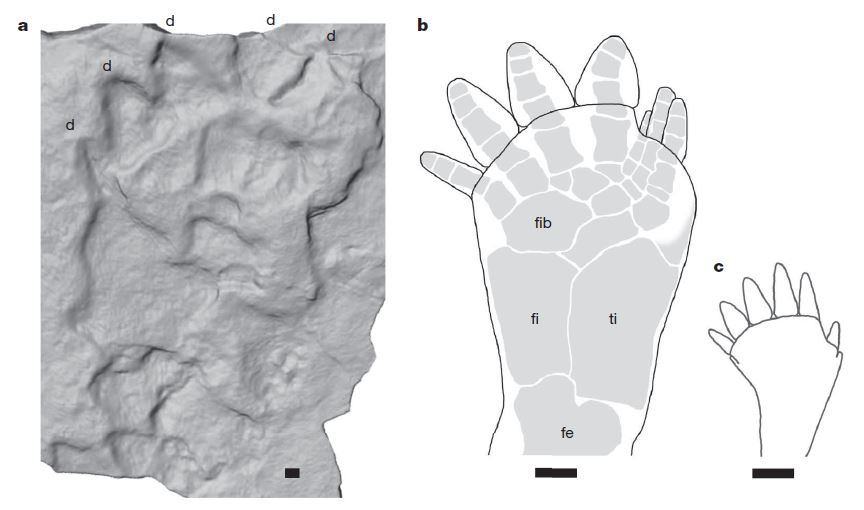
Let’s fast forward to the Mesozoic. Would we necessarily recognize polydactyly in the footprint of a more derived, specialized tetrapod, like a theropod? There are four-toed footprints that are attributed to theropods. Saurexallopus is interpreted to be the footprint of a theropod with four functional toes. The trackmaker was possibly an oviraptorosaur, such as Chirostenotes (Gierlinski and Lockley, 2013). Having a more well-developed digit I compared to other theropods that were running around at the same time (Late Cretaceous) was normal for Chirostenotes and close relatives, so this is a case of anatomy rather than polydactyly.

There are other trackway phenomena that can give the appearance of polydactyly. One of these is a really busy track surface. Busy track surfaces often show animals walking over the footprints of other animals. This often results in dinosaur (and bird) footprints that have the appearance of extra toes, when the simplest explanation is that the footprint is actually one footprint stepping on a different footprint. Another example is when a theropod (usually three-toed) sinks into a substrate deep enough that the hallux and the metatarsus will impress – it gives the appearance of a theropod print with “extra” toes. We see this at the Flatbed Creek Dinosaur Track Site in northeastern British Columbia, where these theropod footprints look like they have five toes instead of the usual three. One toe is indeed a toe – digit I (equivalent to our big toe) – but we don’t usually see that in non-avian theropod footprints. The other “toe” is the impression of the tarsometatarsus.
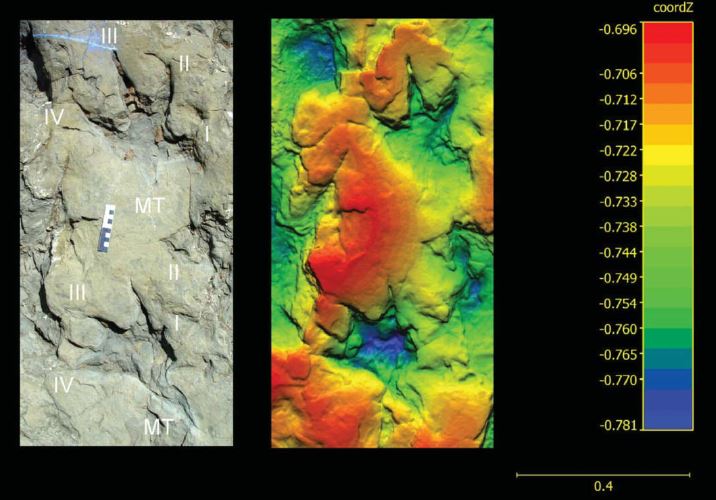
I think that if true polydactyly is to be recognized in theropod footprints, it will have to be in a footprint type that is well-studied and found in many different places, like Eubrontes. This is assuming that archosaurs (crocodiles, dinosaurs, birds) have high enough occurrences of a congenital anomoly like polydactyly in natural populations (in crocodiles it may be related to incubation at extreme temperatures – Google Books link). Polydactyly has also been documented in wild birds: follow this link for a report of polydactyly in a Domestic Pigeon.
Now For the Painful Stuff
You’ve seen some examples of what are not ichnopathologies. Now you get to be rewarded with the really painful-looking footprints and trackways…the ones that you look at and cringe because there is no way those injuries were not extremely uncomfortable. Here I will show the recent additions to the owie-ichnology literature. All of our examples come from non-avian theropods. Much like our modern hawks and eagles, Cretaceous theropods likely used their feet for much more than walking: the feet were also a means of prey capture and restraint (Tanke and Currie, 2000). Theropods led hard, fast lives, and that wear-and-tear showed up on their feet.

Despite all that foot use, wild modern birds of prey have a low occurrence of foot injuries: Bedrosian and St. Pierre (2007) documented a 14% pelvic limb injury rate in Red-tailed Hawks and American Kestrels. Like our modern birds of prey, foot-related injuries are not common in non-avian theropods. The percentage of injured theropod feet is small: Rothschild et al. (2001) observed that healed stress fractures in foot elements range from 0.3% to 6% in large theropods. Other injuries to theropod feet include bony growths that likely resulted from infection/osteomyelitis. So, as these injuries are uncommon in theropod foot bones, we can extrapolate that the resulting footprints from injured feet are uncommon. When we see an ichnopathology, we’re lucky (the trackmaker, however, was less fortunate).
1. Trackway Ichnopathology
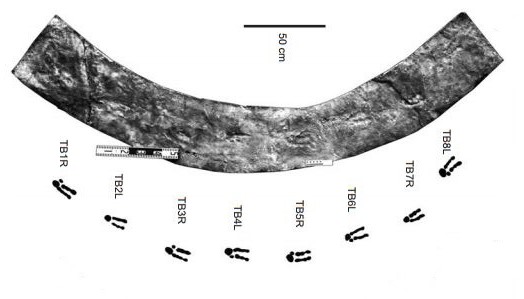
A trackway of a large theropod (cf. Irenesauripus mclearni) from the Early Cretaceous Gates Formation was reported to us. At first, we thought it might be one theropod following right behind another theropod because the steps the animal was taking were WAY too short.

We looked at the substate: it was firm when the animal walked on it, so it wasn’t simply having a tough time slogging through the muck. Then we noticed that the right foot was turned in much more than we usually see in large theropods: non-avian theropods tend to walk with their middle toes pointed roughly straight ahead, or in parallel with the trackway. This theropod was waddling. More specifically, this theropod was using a Pigeon-toed Waddling Gait. It’s hard to say if this gait was the result of an injury to the foot or leg, or if this was a developmental anomaly.
2. Swellings and Dislocations
The most “showy” injuries are those that involve swelling and/or dislocation of a toe. Theropods had no way to reset a dislocated toe, so it would have to walk around with that injury.
Here is a dislocation and swelling-related ichnopathology from the Dakota Group (late Early to early Late Cretaceous) in Colorado.
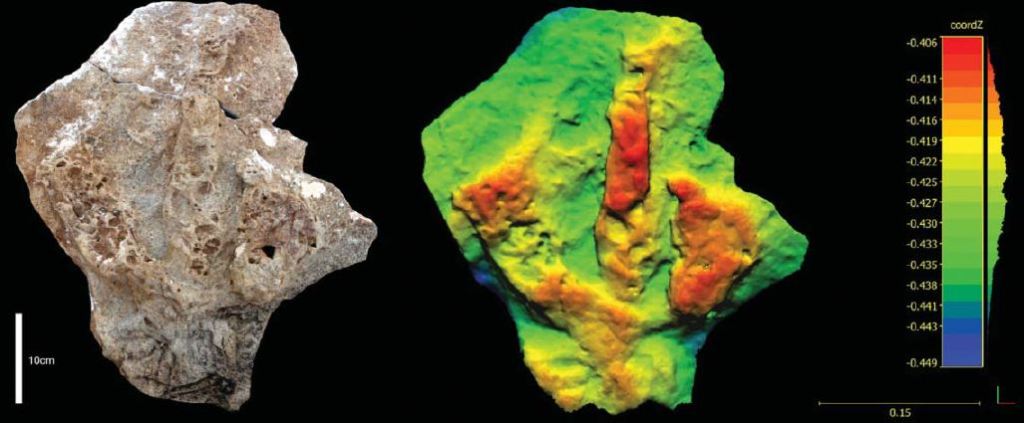
The second most striking dislocation injury I’ve ever seen in a footprint is this large theropod footprint from the Late Cretaceous (approximately 97 million years ago) Kaskapau Formation in northeast British Columbia. Not only is the middle toe (digit III) severely dislocated, but the outer two toes have seemed to compensate for this injury by spreading way out. Unfortunately, the Kaskapau and the Dakota Group pathological footprints were found as singletons. The good news is that, if these animals’ footprints are preserved elsewhere, we have a good chance of linking the footprints to their trackmakers.

Footprint swellings like these are also seen in modern birds. Here is a Canada Goose trackway that I collected a couple of years ago. At the time I made the replica, the toe swelling was hidden by the muddy sediment, but it came out beautifully in the plaster replica.
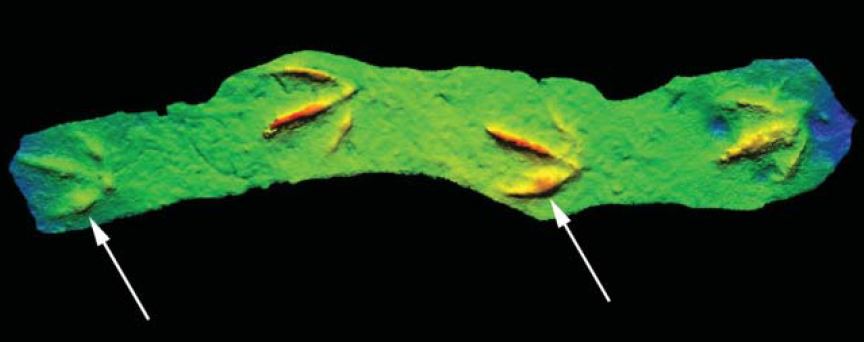
As painful as these two footprints look, they were a mere inconvenience compared to what this next trackmaker must have suffered. Check out this large theropod footprint (first reported by coauthor Darren Tanke) from the Late Cretaceous Wapiti Formation in northwest Alberta.

Yes, you are seeing that correctly: the animal, likely a tyrannosaur (based on the size, and shape of the toe claw, or ungual) stepped on its own toe. Check out how narrow the impression is right before the claw. This could be a trick of the preservation, or it could be that the tissue around the claw was necrotic and beginning to atrophy – this leads to the next level of ichnopathology, also related to tyrannosaurs.
3. Amputations
In 2011 a large theropod trackway consisting of three footprints was reported to us from the B.C. Wapiti Formation. On documenting the trackway, we noticed something peculiar: the inner toe on the left footprints was far too short, while the inner toe on the right footprint was a normal length. Not only did we have the first tyrannosaur trackway preserved, we had one with a rather nasty pathology – a missing toe!
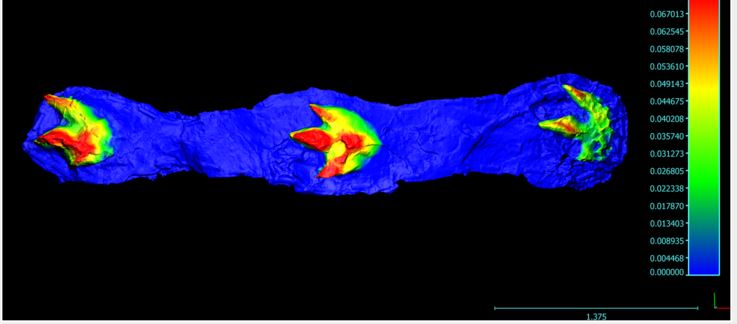
An Ichnopathology Pain Scale
Everyone is familiar with the pain scale used in hospitals. Hospitals are like not allowed to use my favorite pain scale, courtesy of Hyperbole and a Half. Both these pain scales and all of these foot injuries made me ask “What would a theropod pain scale look like?”
So I dusted off my pencils, Googled horrible foot injuries in animals (there are things that I can’t unsee), and used the Bellatoripes fredlundi trackway and all of those horrible swellings and dislocations as inspiration for The Theropod Pain Scale.
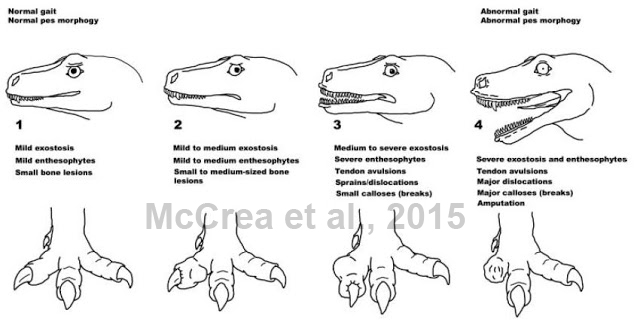
There are two reasons I am immensely proud of this image. First, looking at it made all of my staff simultaneously laugh and cringe in empathy pain for the poor afflicted theropod: apparently the lip quiver did them in (yes, I know the presence of lips is debated in archosaurs – the image was meant to have a touch of comedy in it). Second, it was published! The coauthors liked it, but that didn’t guarantee that the reviewers or the editor would have liked it. I’m glad they did – I do my best teaching and interpretation with humor.
The study of ichnopathologies, just like the study of tracks and traces, gives us a closer look at the complex biological lives of these now-extinct large theropods. Fossilized evidence of injuries reminds us of the fragility and vulnerability of animals often portrayed to the public as rough, tough, indestructible eating machines. Even the most fearsome predator has off days and oopsies. Ichnopathology research also demands that we make use of our living laboratory – outside – as an opportunity to look more closely at the common animal trackways we might take for granted. Each one is an opportunity to learn how an animal’s life is reflected in its footprints.
Owie and Ouchie References
Main paper: McCrea RT, Tanke DH, Buckley LG, Lockley MG,Farlow JO, Xing L, Matthews NA, Helm CW, Pemberton SG, Breithaupt BH (2015) Vertebrate ichnopathology: pathologies inferred from dinosaur tracks and trackways from the Mesozoic, Ichnos, 22:3-4, 235-260
Abel O (1935) Vorzeitliche lebensspuren. Gustav Fisher, Jena.
Baciodonna L, Zucca P, Tommasi L (2010) Posture in ovo as a precursor of footedness in ostriches (Struthio camelus). Behavioural Processes, 83, 130–133.
Bedrosian BE, St. Pierre AM (2007) Frequency of injuries in three raptor species wintering in northeastern Arkansas. Wilson Journal of Ornithology, 119(2), 296–298.
Gierlinski G, Lockley MG (2013) A trackmaker for Saurexallopus: ichnological evidence for oviraptosaurian tracks from the Upper Cretaceous of western North America, p. 526-529 in Titus AL, Loewen MA (eds.) A the top of the Grand Staircase: the Late Cretaceous of southern Utah. Indiana University Press.
Niedzwiedzki G, Szrek P, Narkiewicz K, Narkiewicz M, Ahlberg PE (2010) Tetrapod trackways from the early Middle Devonian period of Poland. Nature 463: doi:10.1038/nature08623
Rothschild BM, Tanke DH, Ford TL (2001) Theropod stress fractures and avulsions as a clue to activity, p. 331–336 in Tanke DH, Carpenter K (eds.). Mesozoic vertebrate life: new research inspired by the paleontology of Philip J. Currie. University of Indiana Press.
Tanke DH, Currie PJ (2000) Head-biting in theropods: paleopathological evidence, in Perez-Moreno BP, Holtz Jr., T, Sanz JL, Moratalla J (eds.). Aspects of theropod paleobiology. Gaia, 15:167–184.
Xing L, Li D, Harris JD, Bell PR, Azuma Y, Fujita M, Lee Y−N, Currie PJ (2013) A new deinonychosaurian track from the Lower Cretaceous Hekou Group, Gansu Province, China. Acta Palaeontologica Polonica 58(4), 723–730.